The Complete Engineering Guide to Molecular Sieve Regeneration
In the highly demanding realms of industrial gas processing, petrochemical refining, and cryogenic air separation, molecular sieves serve as the ultimate line of defense for achieving ultra-high purity standards. These crystalline aluminosilicates, characterized by their uniform microporous structures, are capable of selectively trapping molecules based on precise kinetic diameters and polarities. However, the operational viability, continuous reliability, and economic efficiency of any large-scale adsorption system—whether it is a natural gas dehydration unit, a hydrogen purification skid, or an ethanol dehydration plant—rely entirely on the cyclic, highly controlled physical process known as regeneration.
Without proper and precise regeneration, the internal void spaces of the zeolite crystals rapidly reach their maximum saturation limits. Once saturated, the bed experiences immediate “breakthrough,” allowing contaminants to bypass the adsorbent. This leads to downstream equipment corrosion, catalyst poisoning, pipeline freezing, and catastrophic generation of off-spec products. Understanding the thermodynamics, fluid mechanics, and precise parameter controls behind the regeneration process is not merely an operational recommendation; it is an absolute engineering prerequisite for maintaining plant uptime and safety.
This comprehensive technical guide provides an objective, deep-dive analysis of the physical mechanisms of molecular sieve regeneration. It compares the primary methodologies of Thermal Swing Adsorption (TSA) and Pressure Swing Adsorption (PSA), details standard step-by-step operating procedures, establishes the exact temperature thresholds required across different molecular sieve types (3A, 4A, 5A, and 13X), and examines the root causes of material degradation. By mastering these engineering principles, plant operators and process engineers can optimize system efficiency, vastly extend adsorbent lifespans, and stringently control operational expenditures.
The Core Purpose of Molecular Sieve Regeneration
At the fundamental level of physical chemistry, the adsorption of molecules onto a molecular sieve is an exothermic phenomenon driven by localized electrostatic interactions, dipole-dipole attractions, and Van der Waals forces. When a contaminated gas or liquid stream permeates through the tightly packed adsorbent bed, target molecules (adsorbates) such as water (H₂O), carbon dioxide (CO₂), or hydrogen sulfide (H₂S) are drawn into the crystalline lattice and securely immobilized within the micropores. Over operational time, the available internal surface area and pore volume become fully occupied, establishing a state of thermodynamic equilibrium between the adsorbate remaining in the fluid phase and the adsorbate trapped within the solid matrix.
The primary physical objective of molecular sieve regeneration is to artificially and systematically disrupt this equilibrium. By altering the internal thermodynamic state of the adsorption system—specifically by introducing thermal energy to raise the temperature, or by dropping the total system pressure to decrease the partial pressure of the adsorbate—the kinetic energy of the trapped molecules is elevated. Once this kinetic energy surpasses the activation energy of the binding Van der Waals forces, the adsorbate molecules forcefully detach from the active sites on the aluminosilicate framework and diffuse back into the bulk gas phase. This process is formally defined as desorption.
From an engineering and operational management perspective, the direct, macroscopic purpose of regeneration is the strategic manipulation of the Mass Transfer Zone (MTZ). The MTZ is the specific, active cross-sectional area within the adsorbent bed where the actual mass transfer of contaminants from the fluid stream to the solid zeolite takes place. As the adsorption cycle progresses, the MTZ steadily migrates from the inlet of the vessel toward the outlet. If the leading edge of the MTZ is permitted to reach the bed outlet, contaminant breakthrough occurs.
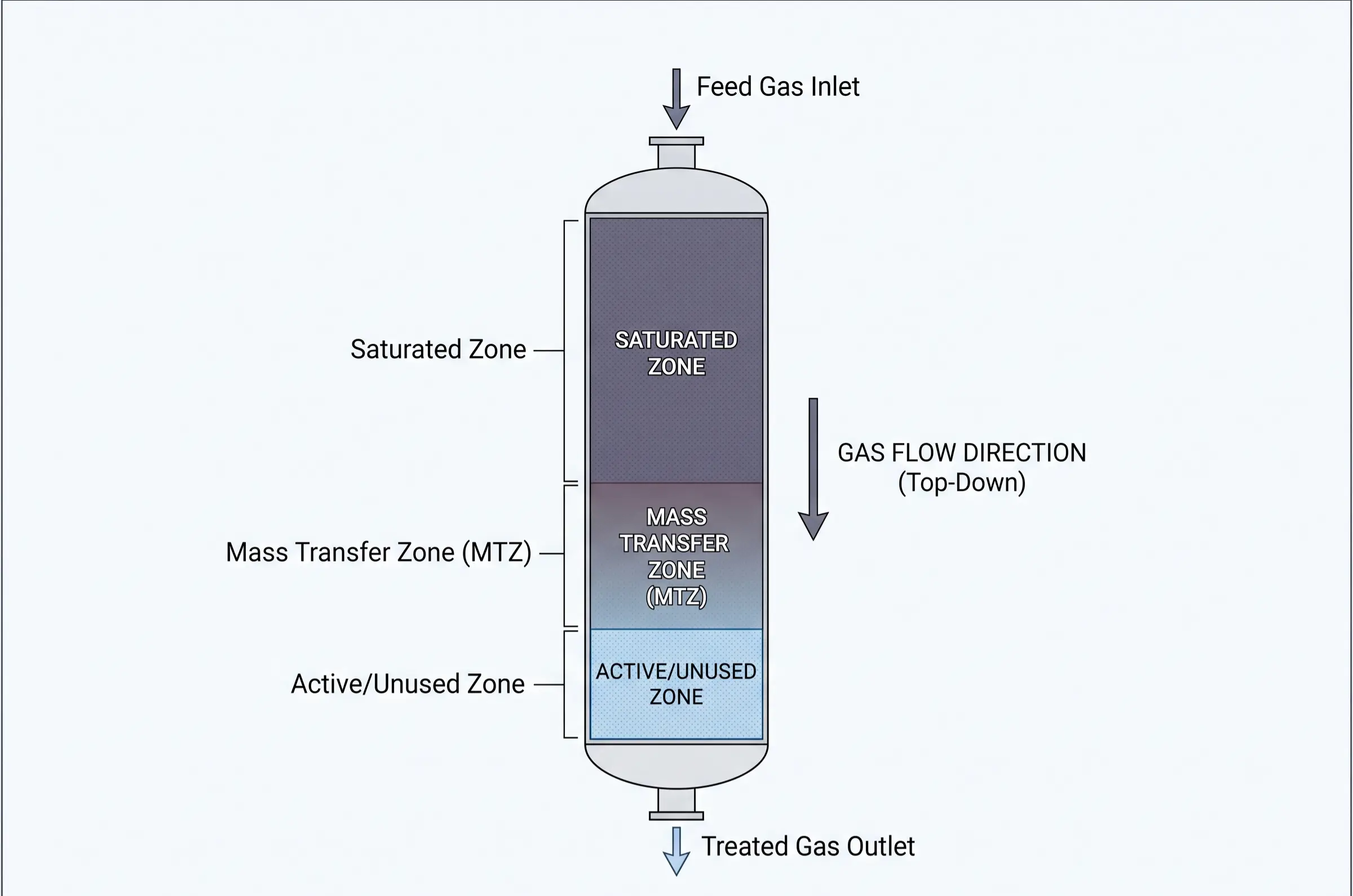
Regeneration is executed to forcefully push the Mass Transfer Zone back to the bottom (or initial inlet) of the bed. By thoroughly stripping the accumulated impurities from the crystalline pores, the dynamic adsorption capacity (the actual working capacity of the bed under specific dynamic flow conditions) is fully restored. Consequently, the ultimate engineering goal of this cyclic restoration is operational expenditure (OPEX) optimization. Highly efficient regeneration allows processing plants to utilize the exact same batch of premium adsorbent inventory for thousands of continuous cycles over several years. This continuously guarantees the absolute purity of downstream process gases while simultaneously preventing the massive capital costs associated with premature adsorbent replacement and the severe energy penalties of inefficient heating.
Molecular Sieve Regeneration vs. Activation: Key Differences
A prevalent and potentially highly damaging misconception within industrial procurement and among junior process engineers is the conflation of the terms “Activation” and “Regeneration.” While both processes involve the application of intense thermal energy to remove volatile substances from the zeolite structure, they occur at entirely different stages of the molecular sieve’s lifecycle, serve fundamentally distinct purposes, and operate under vastly different thermodynamic parameters and temperature constraints. Understanding this distinction is critical for equipment safety.
The Activation Process (Manufacturing Phase)
Activation is a highly controlled, extreme-temperature metallurgical process that occurs exclusively within the molecular sieve manufacturer’s production facility, typically inside massive industrial rotary kilns or specialized roasting furnaces. When raw synthetic zeolite is first crystallized during chemical manufacturing, its microscopic pores are completely flooded with water of crystallization and the organic template agents (such as amines or specific quaternary ammonium salts) that were utilized to direct the formation of the specific crystal framework. In this “raw” and uncalcined state, the molecular sieve possesses absolutely zero dynamic adsorption capacity.
To render the material functionally porous, manufacturers subject the raw powder or formed pellets to extreme activation temperatures ranging from 500°C to 600°C (932°F to 1112°F). At these extreme thermal thresholds, the organic templating molecules undergo complete thermal decomposition and combustion, while the deep-seated structural water is violently expelled from the crystalline lattice. This process creates the vast, empty, three-dimensional void spaces that will eventually trap industrial contaminants. Because activation is a one-time formatting process designed to clear the initial chemical scaffolding, the thermal limits applied here push the absolute physical tolerance of the material. For operators looking to understand the complex manufacturing steps behind this initial preparation, we highly recommend reading a complete guide on how to activate molecular sieve to grasp the foundational material science.
The Regeneration Process (Operational Phase)
In stark contrast, Regeneration occurs at the end-user’s facility, taking place entirely within the localized environment of the operational field equipment, such as the twin-tower dehydration vessels in a natural gas processing plant or the pre-purification units (PPU) in a cryogenic air separation plant. Regeneration is a cyclic, routine maintenance procedure designed exclusively to remove the specific process impurities (like atmospheric moisture, trace CO₂, or sulfur compounds) that have accumulated during standard plant operation.
The operational parameters for field regeneration are strictly bounded by established material science limitations. Standard industrial regeneration temperatures are meticulously controlled between 150°C and 350°C (302°F to 662°F), depending entirely on the type of molecular sieve and the tenacity of the specific adsorbed contaminant. It is an absolute, non-negotiable engineering redline that localized bed temperatures during field regeneration must never exceed 450°C (842°F). Exposing an operational molecular sieve bed to temperatures approaching or exceeding 450°C triggers a permanent phase transition in the aluminosilicate binder and the zeolite crystals themselves, leading to irreversible crystal lattice collapse, widespread pore closure, and the total, permanent loss of dynamic adsorption capacity.
Primary Methods for Molecular Sieve Regeneration
Industrial adsorption systems are fundamentally categorized by the primary thermodynamic mechanism utilized to reverse the adsorption equilibrium. The two most dominant and technologically mature methodologies employed globally are Thermal Swing Adsorption (TSA) and Pressure Swing Adsorption (PSA). The engineering selection between these two routes is dictated by the required purity of the product gas, the volume of gas being processed, the specific concentration of the impurities, and the availability of plant utilities such as waste heat or high-pressure compression capabilities.
Thermal Swing Adsorption (TSA) Process
The Thermal Swing Adsorption (TSA) process is architected upon the thermodynamic principle that the adsorption capacity of a molecular sieve decreases exponentially as the systemic temperature increases. At ambient operating temperatures (e.g., 20°C to 40°C), the zeolite lattice exhibits a massive affinity for polar molecules and trace contaminants. However, by introducing a high-temperature purge gas into the bed during the regeneration phase, the localized thermal energy drastically increases the kinetic vibration of the adsorbed molecules. This added thermal energy easily overcomes the electrostatic holding forces, driving the equilibrium aggressively toward the gas phase for desorption.
TSA systems are universally favored for operational scenarios requiring extreme, ultra-trace removal of impurities, commonly referred to as “deep purification.” This includes cryogenic natural gas dehydration (preventing hydrate formation at sub-zero temperatures) and deep desulfurization or decarbonization. Operationally, TSA is characterized by long, sustained cycle times, typically ranging from 8 to 24 hours per cycle, because heating and cooling massive beds of solid zeolite is a slow, thermally intensive process. Consequently, TSA strictly requires a robust external heat source, such as electric immersion heaters, direct-fired gas heaters, or integrated waste heat recovery systems.
Pressure Swing Adsorption (PSA) Process
Conversely, the Pressure Swing Adsorption (PSA) process operates on the dynamic kinetic principle that adsorption capacity is highly dependent on the partial pressure of the target gas. According to Langmuir adsorption isotherm principles, at elevated systemic pressures, the molecular sieve rapidly adsorbs large quantities of gas. When that pressure is rapidly reduced, the equilibrium shifts instantaneously, causing the adsorbate molecules to desorb and detach from the solid matrix.
PSA technology is the industry standard for large-volume, macroscopic gas separation applications rather than trace impurity removal. Typical applications include the generation of high-purity industrial oxygen or nitrogen from atmospheric air, or the extraction of ultra-pure hydrogen from synthesis gas or refinery off-gases. Operationally, PSA is distinct because it functions entirely at near-ambient temperatures. The thermal swing is negligible. Instead, the process relies heavily on rapid, continuous mechanical cycling. PSA cycle times are extremely short, often ranging from just a few minutes down to mere tens of seconds. The entire regenerative force relies on the instantaneous release of system pressure (blowdown), which dynamically ejects the contaminants from the micropores without the need for external thermal heating.
The Standard Molecular Sieve Regeneration Process
To fully grasp the complexities of molecular sieve management, it is necessary to examine the precise, step-by-step operating procedures that govern these systems. In modern industrial control rooms, TSA and PSA systems represent entirely different operational paradigms. Below is a detailed breakdown of the standard regeneration sequences for both methodologies, highlighting the parallel nature of thermal versus kinetic desorption mechanisms.
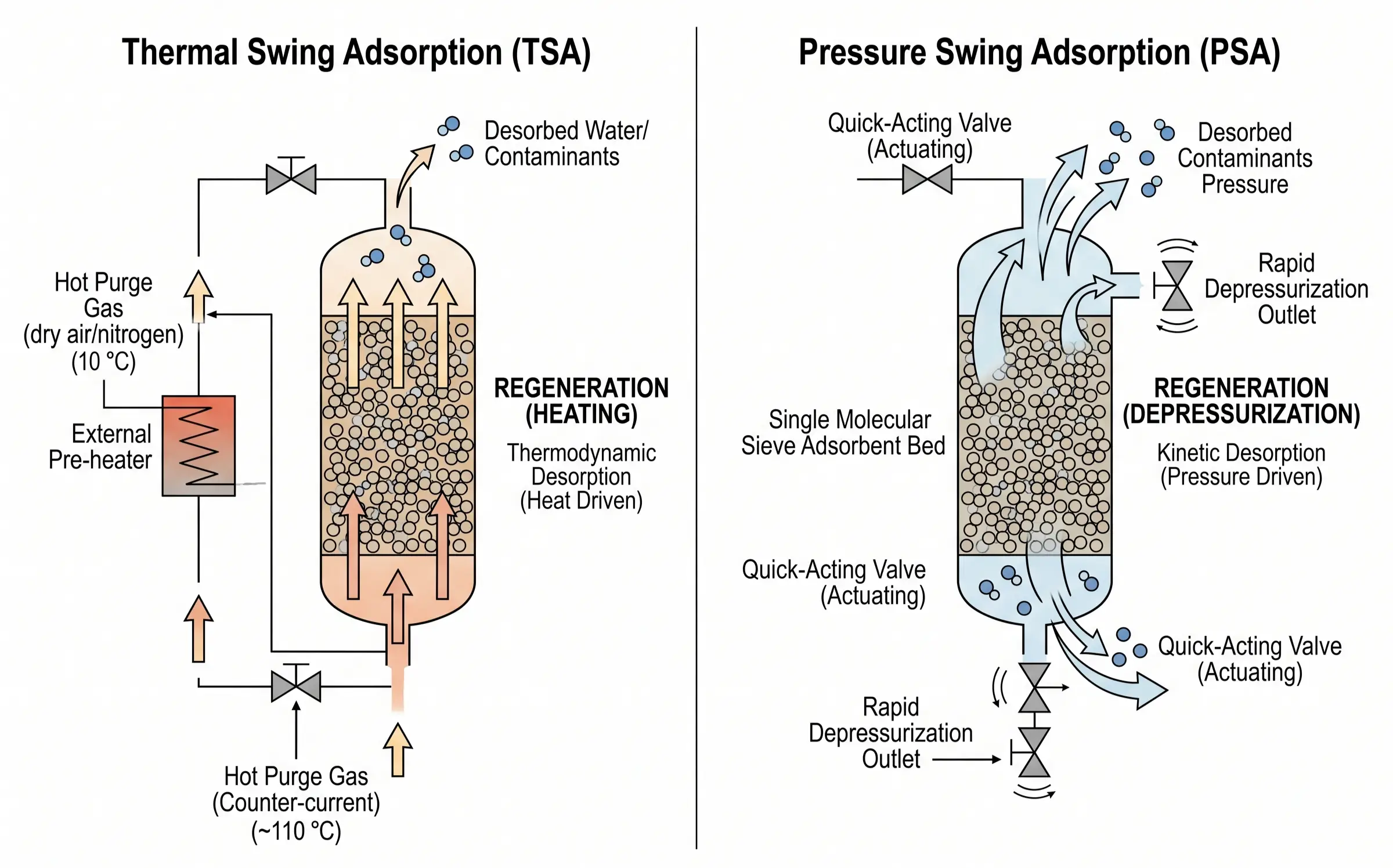
Standard TSA Regeneration Process (Time-based Sequence)
The TSA sequence is a meticulously timed thermal procedure designed to safely import and export massive amounts of heat without damaging the fragile ceramic structure of the adsorbent.
Phase 1: Heating with Purge Gas (Heating & Desorption)
In the physical reality of a TSA system, heating and sweeping (purging) are not sequential steps; they are intrinsically parallel and coupled mechanisms. During Phase 1, a heavily dried, high-temperature regeneration gas (the purge gas) is introduced into the saturated bed. Engineering best practice mandates that this hot gas flow in a Counter-current direction relative to the normal adsorption flow. If adsorption occurs top-down, heating must occur bottom-up. This prevents the desorbed water from the highly saturated inlet zone from being pushed deeper into the ultra-dry outlet zone of the bed, which would permanently poison the polishing section of the sieve.
During this phase, the thermodynamic action of the rising temperature breaks the Van der Waals forces, while the kinetic action of the constant sweep gas continuously lowers the vapor pressure of the contaminant in the surrounding void space, physically carrying the liberated molecules out of the vessel. To prevent catastrophic structural failure, operators must strictly control the ramp-up rate. The heating rate is typically restricted to 30°C to 50°C per hour. Rapid thermal spikes cause immense thermal gradients between the exterior and interior of the zeolite pellets, leading to thermal stress fracturing. The heating phase is considered complete only when the effluent gas temperature at the outlet of the bed reaches a minimum of 150°C (though often higher depending on the sieve) and maintains a stable temperature plateau for several hours, proving that all deep-seated latent water has been successfully boiled off and evacuated.
Phase 2: Cooling with Dry Sweep Gas
Once desorption is complete, external heat is secured, but the bed is currently sitting at 200°C to 300°C. It cannot be placed back online to process ambient gas, as its adsorption capacity at elevated temperatures is virtually zero. Phase 2 involves continuing the flow of the purge gas, but with the heater turned off, slowly bringing the bed temperature down safely to ambient levels.
This phase contains the most critical, high-risk operational redlines in the entire process. The cooling gas utilized must have an exceptionally low dew point (e.g., lower than -40°C). It is strictly forbidden to use wet, untreated process gas to cool a hot molecular sieve bed. If a hot, completely desiccated zeolite is suddenly exposed to moisture, the crystals will instantaneously adsorb the water. This rapid adsorption releases a massive, violent burst of thermal energy known as the Latent Heat of Adsorption. This localized energy release can cause bed temperatures to spike uncontrollably by hundreds of degrees in seconds, literally cooking the binder, boiling the trapped water internally, and shattering the pellets into useless dust (powdering), effectively destroying the entire multi-million dollar inventory in a single error.
Standard PSA Regeneration Process (Pressure-based Sequence)
The PSA sequence eschews heat entirely, relying instead on high-speed mechanical valves and the kinetic forces of rapid depressurization to strip contaminants from the bed.
Phase 1: Depressurization / Blowdown
When the online adsorber vessel reaches its calculated saturation point, the inlet feed valves rapidly close. Immediately, exhaust valves open, causing the internal vessel pressure to plummet from its high working pressure down to near atmospheric pressure (or a slight positive pressure). According to the adsorption isotherms, this sudden, drastic drop in partial pressure alters the equilibrium state instantaneously. The massive volume of adsorbates (such as nitrogen in an oxygen generator) is forcibly expelled from the micropores and exhausted into the atmosphere or a secondary recovery system.
The engineering redline here revolves around fluid dynamics. The rate of depressurization (dP/dt) must be strictly controlled by orifice plates or tuning valves. If the blowdown is too violent, the extreme upward velocity of the expanding gas will physically lift the adsorbent beads, causing the bed to “fluidize” or boil. This violent tumbling and friction between the ceramic pellets generates massive amounts of abrasive dust, leading to severe pressure drop issues and eventual mechanical failure of the system.
Phase 2: Purging / Sweeping
While the vessel sits at its lowest pressure state, mere depressurization is rarely sufficient to clean the deepest micropores. To achieve total regeneration, a small slipstream of ultra-high purity product gas (or another highly dried carrier gas) is introduced at low pressure, flowing in a counter-current direction. This sweeping action aggressively scrubs the remaining trace contaminants out of the macropores and interstitial spaces, breaking localized equilibrium and completely resetting the dynamic adsorption capacity of the bed to its maximum baseline.
Phase 3: Repressurization
Before the vessel can be brought back online to accept high-pressure feed gas, it must be repressurized safely. Opening a high-pressure feed valve into a low-pressure vessel would create a sonic shockwave, pulverizing the top layer of the molecular sieve bed. Therefore, repressurization is achieved smoothly, often utilizing product gas from a parallel active tower (a process known as pressure equalization). This not only conserves valuable pressurized gas, increasing overall plant efficiency, but also ensures the bed pressure is smoothly equalized to the working pressure, ready to seamlessly begin the next adsorption cycle without mechanical shock.
Regeneration Parameters Across Different Molecular Sieve Types
Because molecular sieves are synthesized with varying pore diameters and framework compositions, their specific affinity for water and other polar molecules differs significantly. Consequently, the thermodynamic energy required to break the adsorption bonds during a TSA cycle must be carefully calibrated to the specific type of zeolite installed in the vessel. Applying a generic heating profile to all molecular sieves will result in either incomplete desorption (if the temperature is too low) or accelerated hydrothermal degradation (if the temperature is unnecessarily high).
The following technical matrix outlines the optimal regeneration parameters, core operational difficulties, and critical temperature redlines for the four most common industrial molecular sieve variants.
| Sieve Type | Optimal Temp Range | Core Difficulty / Application Notes | Critical Operation Point |
|---|---|---|---|
| 3A | 170°C – 220°C | Due to its small pore size (3 Ångströms), 3A is primarily used in ethanol dehydration and cracked gas (ethylene/propylene) drying. The core difficulty is preventing co-adsorption of reactive olefins. Temperatures must be kept strictly within range; excessive heat in the presence of organics can lead to severe coking and carbon deposition on the exterior of the beads. | Do not exceed 250°C to avoid accelerating polymer formation and coking. |
| 4A | 200°C – 250°C | The standard workhorse for general instrument air drying and natural gas dehydration. Desorption is relatively straightforward, but the bed must be thoroughly purged to ensure dew points of -100°C can be achieved upon returning to service. | Maintain steady heating ramp rates. Exceeding 450°C will permanently collapse the Sodium A crystal structure. |
| 5A | 200°C – 300°C | Often utilized for the separation of normal and isoparaffins, as well as desulfurization (H₂S removal). The divalent Calcium ions create a stronger electrostatic field than Sodium, meaning contaminants are held more tightly. Consequently, the resistance to desorption is noticeably higher than in 4A, necessitating elevated baseline temperatures to achieve complete regeneration. | Careful monitoring of effluent gas required to ensure complete sulfur desorption before cooling begins. |
| 13X | 250°C – 350°C | Highest Desorption Energy Required. 13X is unique; while it has the largest pore diameter (10 Ångströms), the physical reason it requires the highest regeneration temperature is fundamentally due to its exceptionally low Silica-to-Alumina (Si/Al) ratio. This low ratio results in a highly dense, intensely strong electrostatic field and extreme polar affinity within the crystal framework. The latent heat of adsorption for water on 13X is immense. Therefore, operators must provide substantially higher thermal energy to break this powerful equilibrium. | Extended temperature plateau required. |
Mechanisms of Molecular Sieve Degradation and Aging
No molecular sieve lasts forever. Even under the most meticulous operational control, the dynamic adsorption capacity of a zeolite bed will experience a steady decline over thousands of regeneration cycles. However, understanding the physical and chemical mechanisms of this degradation separates normal wear-and-tear from premature, catastrophic failure. For process engineers, mitigating these failure modes is the essence of lifecycle management.
Hydrothermal Aging (Dealumination)
Hydrothermal aging is the most pervasive and unavoidable form of chemical degradation in TSA systems. During the early stages of the heating phase, if the bed still contains a massive volume of trapped water, the localized application of heat causes this water to vaporize into high-temperature, high-pressure steam. When the delicate aluminosilicate lattice is exposed to high-temperature steam over thousands of hours, a chemical reaction known as dealumination occurs. The steam chemically attacks the framework, stripping aluminum atoms out of the crystalline structure. This causes the internal micropores to gradually collapse and fuse together, permanently reducing the total internal surface area and permanently shrinking the dynamic capacity. To mitigate this, operators must ensure that the regeneration purge gas is exceptionally dry and that heating ramp rates are controlled to allow water to evacuate before systemic temperatures reach their peak.
Coking and Contamination (Pore Blockage)
Unlike hydrothermal aging, which damages the structure, coking entirely blocks access to it. If the incoming process gas or the regeneration carrier gas contains trace amounts of heavy hydrocarbons, compressor lubricating oils, or reactive amines, disaster is imminent. As the bed is heated to 200°C – 300°C during regeneration, these heavy organic molecules undergo thermal cracking and carbonization directly on the surface of the zeolite pellets. This forms a hard, impenetrable layer of carbon (coke) that acts like cement, permanently sealing the entrances to the micropores. Even though the internal capacity remains technically intact, the adsorbate molecules can no longer enter the pores. Preventing this requires absolute vigilance upstream, mandating the installation and rigorous maintenance of high-efficiency coalescing filters and carbon guard beds to trap oils before they ever touch the molecular sieve.

Thermal Stress and Mechanical Powdering
Mechanical failure manifests as “dusting” or “powdering,” leading to severe pressure drop spikes across the bed. Zeolite pellets are ceramics; they are exceptionally strong under static compression but highly brittle when subjected to tension or thermal shock. If an operator applies heat too rapidly, the exterior of the pellet expands thermally while the core remains cool, generating immense internal shearing stress that tears the pellet apart from the inside. Similarly, if gas velocities during the PSA blowdown phase or TSA purge phase exceed design limits, the aerodynamic lift forces will cause the pellets to physically bounce, grind, and abrade against one another. To prevent this mechanical destruction, strict adherence to step-by-step temperature ramp rates, controlled valve sequencing, and proper bed support/compaction protocols must be enforced at all times.
Performance Evaluation and Efficiency Factors
Theoretical knowledge of regeneration must translate into actionable, measurable diagnostics on the plant floor. Operators must continuously evaluate the efficiency of the regeneration cycle using real-time telemetry data from the Distributed Control System (DCS). Relying solely on a timer to dictate cycle completion is a rudimentary and high-risk operational strategy.
Key Indicators of a Successful Regeneration
- Temperature Profile Convergence: The most reliable thermodynamic indicator of a successful TSA heating phase is the narrowing of the temperature differential between the bed inlet and outlet. At the beginning of heating, the hot gas loses its energy entirely to the cold bed and evaporating water. As the bed dries and heats up, the thermal wave pushes through to the bottom. When the effluent (outlet) temperature rises, reaches the target threshold (e.g., 150°C+) and stabilizes, it proves the bed has reached thermal equilibrium and the latent water is gone.
- Online Dew Point Stability: The ultimate performance metric. Upon switching the regenerated vessel back to the active adsorption cycle, the moisture analyzer on the product gas line should immediately register an ultra-low, stable dew point (often below -100°C depending on the system). If the dew point spikes early in the cycle, it is the primary indicator of an incomplete regeneration or a severely degraded bed.
- Pressure Drop (ΔP) Integrity: Mechanical integrity is monitored via differential pressure transmitters across the bed. A healthy bed maintains a stable, predictable pressure drop dictated by gas velocity. If the ΔP begins a steady, irreversible upward trend over several cycles, it is a definitive warning sign that the pellets are powdering, breaking down, and clogging the interstitial gas flow channels, demanding immediate engineering investigation.
Critical Variables Impacting Regeneration Efficiency
When troubleshooting an inefficient regeneration cycle, process engineers must audit three fundamental variables:
- Purge Gas Volumetric Flow Rate: Thermodynamics requires heat, but kinetics requires flow. If the mass flow rate of the regeneration carrier gas is too low, it will lack the carrying capacity to sweep the desorbed moisture out of the vessel. The moisture will simply reach localized equilibrium and redeposit deeper in the bed as it cools.
- Heater Power and Heat Transfer Efficiency: Over time, electric heater bundles can scale, or fired heaters can lose burner efficiency. If the required thermal wattage is not being successfully transferred into the purge gas, the bed will never reach the critical desorption temperatures, regardless of how long the cycle runs.
- Valve Sealing and Internal Leakage: This is the most common, yet most hidden, culprit. Large switching valves inevitably wear down. If a hot regeneration valve leaks even a fractional amount of wet feed gas into the system during the cooling or standby phase, the bed will prematurely pre-load with moisture before the adsorption cycle even begins, drastically shortening the operational cycle time.
Selecting High-Durability Molecular Sieves
While precise regeneration parameters mitigate degradation, the innate mechanical and hydrothermal stability of the molecular sieve ultimately dictates the system’s lifespan. Inferior adsorbents are highly susceptible to dusting and structural collapse under continuous thermal and pressure swings.
JALON, a leading manufacturer with over 22 years of expertise and a 55,000-ton annual capacity, engineers molecular sieves designed for extreme cyclic stress. Supported by an expert team with 20+ years of on-site petrochemical experience, we provide fully customized adsorption solutions and free technical evaluations to optimize your regeneration process and minimize unplanned downtime.
Frequently Asked Questions on Sieve Lifespan
There is no universal standard for the exact number of cycles, as lifespan is dictated entirely by the severity of the operational environment. In highly pristine, pure, and dry gas applications—where the feed gas contains no reactive hydrocarbons or heavy contaminants—a high-quality molecular sieve can easily endure several thousand regeneration cycles, yielding an operational lifespan of 3 to 5 years. However, in aggressive, harsh conditions involving heavy oils, acid gases, or frequent extreme thermal shocks, the crystalline structure will degrade much faster, potentially reducing the viable lifespan to 12 to 18 months before a total replacement is required to maintain product purity.
Failing to fully regenerate the bed triggers a cascading operational failure loop. If trace moisture or contaminants are left behind in the micropores due to insufficient temperature or inadequate purge flow, the available adsorption capacity for the next cycle is mathematically reduced. Consequently, the bed will reach breakthrough much faster, forcing the automation system to shorten the cycle time. This leads to wildly increased valve switching frequencies, increased energy consumption for more frequent heating, accelerated hydrothermal aging of the remaining healthy zeolite, and an exponential increase in the risk of off-spec product reaching downstream equipment, ultimately leading to highly expensive, unplanned plant shutdowns.









