This Guide Details Zeolite Catalyst Types, Applications, and Selection
In the modern chemical processing and refining industries, the pursuit of higher yields, stricter environmental compliance, and optimized energy consumption has driven catalyst engineering to unprecedented levels of sophistication. At the very core of this industrial evolution lies a remarkable class of materials: zeolite catalysts. These advanced materials serve as the unyielding backbone for processes ranging from massive crude oil fluid catalytic cracking (FCC) to highly precise emission control systems. This comprehensive technical guide details the fundamental physical and chemical nature of zeolite catalysts, systematically breaks down their core classification dimensions, and explores their critical applications across petrochemical, environmental, and fine chemical sectors. Furthermore, it provides engineers and procurement specialists with a strategic framework for catalyst selection, alongside actionable solutions to overcome inherent technical bottlenecks such as mass transfer limitations and deactivation. By understanding the complete lifecycle management of these catalysts, plant operators can significantly enhance their return on investment (ROI) and maintain continuous, efficient production.
What Are Zeolite Catalysts?
At their fundamental level, zeolite catalysts are highly crystalline, three-dimensional aluminosilicate materials. They are meticulously structured networks composed of corner-sharing silicon-oxygen (SiO4) and aluminum-oxygen (AlO4) tetrahedra. The defining characteristic of a zeolite catalyst lies in its dual identity: it operates simultaneously as a potent "solid acid" and a highly precise molecular sieve catalyst. When a tetravalent silicon ion (Si4+) in the crystal lattice is isomorphously substituted by a trivalent aluminum ion (Al3+), it creates a localized net negative charge in the framework. To maintain electrical neutrality, this charge must be balanced by a non-framework cation. When this compensating cation is a proton (H+), it forms a Brønsted acid site. This unique structural chemistry endows zeolite catalysts with three core physical and chemical features that dictate their immense industrial value:
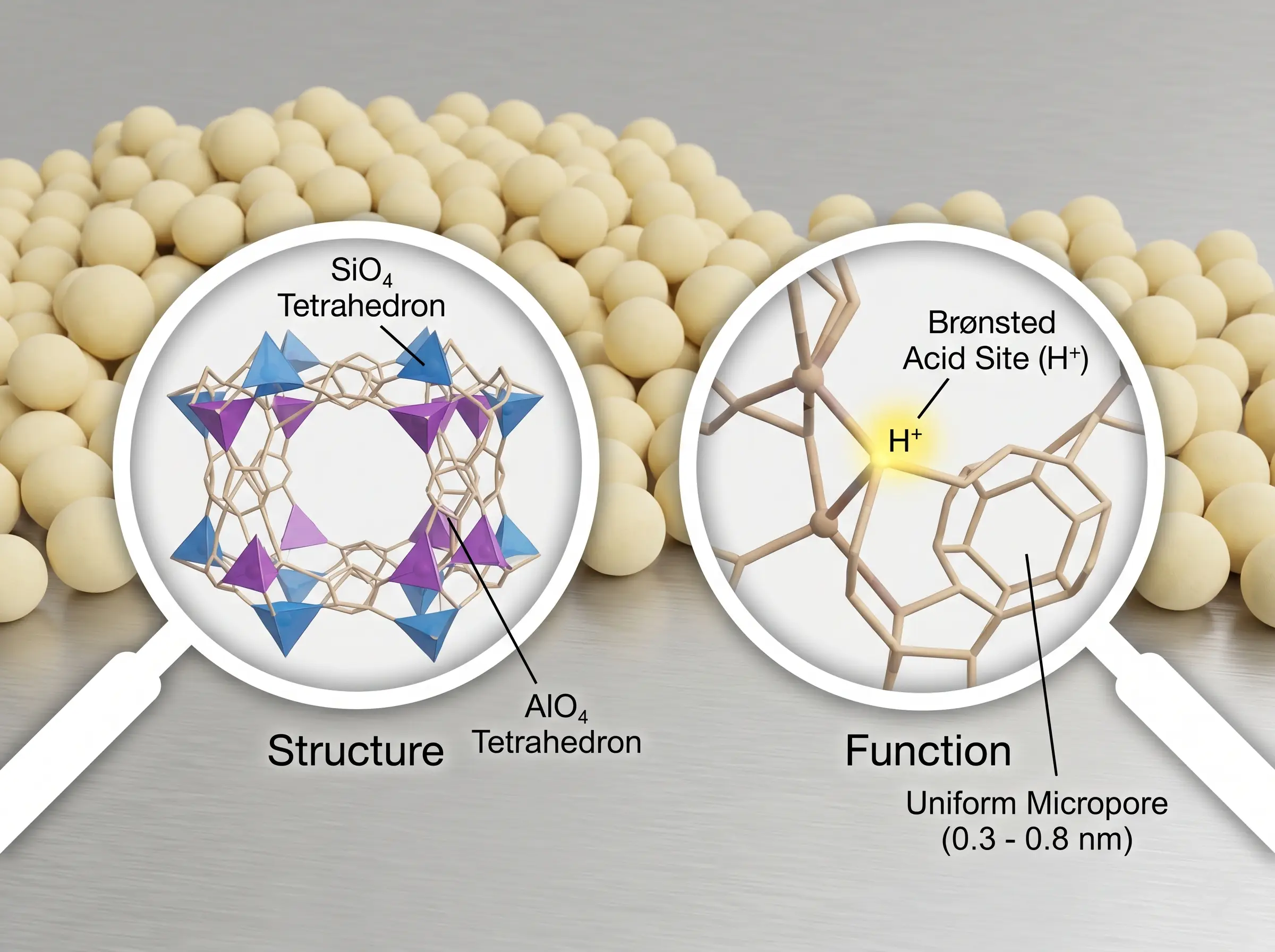
- Uniform Microporous Structure: Unlike amorphous catalysts with a random distribution of pore sizes, zeolites feature an extensively ordered crystalline pore network (typically 0.3 to 0.8 nm). This architecture precisely matches the kinetic diameters of many petrochemical molecules, enabling strict "shape-selective catalysis" that tightly controls reactant ingress, product egress, and transition-state formation.
- Extremely High Surface Area: The intricate internal labyrinth of channels and cages provides a massive internal surface area, routinely exceeding 500 to 1,000 square meters per gram. This extreme surface-to-volume ratio guarantees an exceptionally high concentration of active reaction sites, translating to significantly higher conversion rates in industrial reactors.
- Highly Tunable Acidic Sites: The catalytic potency of a zeolite is actively engineered. The acidity—both in total acid site density and acid strength—can be precisely tailored by altering the silicon-to-aluminum (Si/Al) ratio. This allows chemical engineers to customize the catalyst to match the exact thermodynamic and kinetic requirements of target reactions.
Classification Dimensions and Core Zeolite Models
Given the vast array of synthetic zeolite materials available in the industrial market, selecting the appropriate catalyst requires a systematic understanding of their structural and chemical variations. Zeolite catalysts are typically evaluated and classified across four key technical dimensions, which ultimately dictate their thermal stability, chemical behavior, and specific industrial applicability.
Four Key Dimensions of Zeolite Classification
- Pore Size and Ring Architecture: Zeolites are primarily categorized by the size of their pore openings, which are determined by the number of oxygen atoms forming the aperture ring. Small-pore zeolites (e.g., 8-membered rings) have pore diameters ranging from roughly 0.3 to 0.45 nm, allowing only highly linear molecules to pass. Medium-pore zeolites (e.g., 10-membered rings), with diameters between 0.45 and 0.6 nm, are the workhorses of shape-selective aromatics processing. Large-pore zeolites (e.g., 12-membered rings) feature openings of 0.6 to 0.8 nm, making them essential for processing bulkier molecules found in heavy gas oils. Recent advancements have also introduced mesoporous zeolites, integrating pores larger than 2 nm to facilitate the transport of extremely large molecules.
- Silicon-to-Aluminum Ratio (Si/Al Ratio): The Si/Al ratio is a critical parameter that dictates the material's hydrophobicity, acid density, and structural durability. Low-silica zeolites (Si/Al ratio of 1 to 1.5) possess a very high concentration of aluminum, resulting in dense acid sites and extreme hydrophilicity (water affinity); however, they lack thermal stability. Medium-silica zeolites (Si/Al ratio of 2 to 5) offer a balance of properties. High-silica zeolites (Si/Al ratio greater than 10, extending up to pure silica frameworks) are highly hydrophobic and exhibit exceptional hydrothermal stability, making them robust enough to withstand severe industrial regeneration conditions involving high-temperature steam.
- Chemical Composition: While standard zeolites are pure aluminosilicates, advanced engineering has allowed for the isomorphous substitution of framework atoms with various heteroatoms to alter catalytic behavior. For example, incorporating titanium into the framework produces Titanium Silicalite-1 (TS-1). TS-1 exhibits unique catalytic oxidation properties, utilizing hydrogen peroxide as an environmentally benign oxidant for the epoxidation of olefins and the hydroxylation of aromatics, independent of traditional strong acid catalysis.
- Pore Dimensionality: The internal channel architecture heavily influences how molecules travel through the catalyst bed. One-dimensional (1D) channel systems force molecules to move in a single file; if a heavy byproduct forms and blocks the channel, the entire pore becomes deactivated. Two-dimensional (2D) and three-dimensional (3D) channel networks provide intersecting pathways. A 3D architecture allows reactant and product molecules to bypass localized blockages, drastically improving the catalyst's resistance to deactivation by coking and extending its operational lifespan.
Essential Zeolite Models and Nomenclature
Understanding the specific types of zeolite catalyst and their corresponding International Zeolite Association (IZA) framework codes is essential for proper specification. Below is an objective comparison of the most critical industrial zeolite models.
| Zeolite Model Name | Topology Code (IZA) | Pore Characteristics | Core Industrial Applications |
|---|---|---|---|
| Zeolite Y (Faujasite) | FAU | Large pore (12-ring, ~0.74 nm), 3D intersecting channels with large supercages (~1.3 nm) | Fluid Catalytic Cracking (FCC), Hydrocracking (processing heavy vacuum gas oils) |
| ZSM-5 | MFI | Medium pore (10-ring, ~0.51 x 0.55 nm), 3D intersecting channels | Xylene isomerization, Methanol-to-Gasoline (MTG), Toluene disproportionation |
| Zeolite Beta | BEA | Large pore (12-ring, ~0.66 x 0.67 nm), complex 3D interconnected pore system | Aromatics alkylation (e.g., cumene and ethylbenzene production), advanced hydrocracking |
| SAPO-34 | CHA | Small pore (8-ring, ~0.38 nm), 3D chabazite cage structure | Methanol-to-Olefins (MTO), advanced automotive emission control (NH3-SCR) |
| Mordenite | MOR | Large pore (12-ring, ~0.65 x 0.70 nm), primarily 1D channel system | Light naphtha isomerization, selective alkylation of biphenyl |
Decoding Zeolite Nomenclature: Industrial specifications often present complex alphanumeric codes. Taking the commercial model HZSM-5 (Si/Al=30) as an example, the nomenclature can be systematically decoded. The prefix "H" indicates the protonated form, meaning the cation exchange sites are occupied by hydrogen protons, confirming the material is currently in its active, solid acid state (as opposed to a Na-ZSM-5 precursor). "ZSM-5" stands for Zeolite Socony Mobil-5, representing the specific framework structure (MFI topology). The suffix "(Si/Al=30)" explicitly states the molar ratio of silicon to aluminum in the framework, indicating a highly siliceous, hydrothermally stable variant with strong, isolated acid sites. Another prevalent example is USY, which stands for Ultra-Stable Y. This designation implies that a standard Zeolite Y has undergone severe hydrothermal treatment and chemical dealumination to remove framework aluminum, thereby drastically increasing its high-temperature stability for harsh fluidization environments.
Prime Industrial Applications of Zeolite Catalysts
When evaluating diverse zeolites uses, their profound economic impact is most visible across four macro-industrial sectors. Their ability to conduct precise molecular transformations at massive scales has fundamentally shaped modern energy and material supply chains.

Oil Refining and Petrochemical Processing
- Fluid Catalytic Cracking (FCC): FCC represents the most critical and economically significant secondary processing operation within a modern petroleum refinery. It is crucial to note that an industrial-grade FCC catalyst is not merely raw zeolite powder. It is a highly engineered microspherical composite. It utilizes 10% to 50% of USY (Ultra-Stable Y) zeolite as the core active catalytic engine. This zeolite is encapsulated within an active matrix (often alumina, which provides preliminary pre-cracking of massive hydrocarbon molecules), a filler (typically inert kaolin clay for thermal mass), and an inorganic binder. This mixture is spray-dried to form highly robust microspheres ranging precisely from 60 to 75 microns in diameter. This specific size range and spherical morphology are absolute prerequisites for fulfilling the rigorous aerodynamic fluidization requirements within the high-velocity riser reactor. Here, the USY zeolite efficiently cracks heavy, low-value vacuum gas oils into high-octane gasoline, diesel blending components, and valuable low-carbon olefins like propylene and butylene.
- Hydrocracking: This process synergizes catalytic cracking with high-pressure hydrogen addition to upgrade the heaviest crude fractions. Hydrocracking catalysts are bifunctional; they utilize a modified Zeolite Y or Zeolite Beta as a catalyst support to provide the necessary solid Brønsted acidity for carbon-carbon bond cleavage, while incorporating supported noble or transition metals (like Platinum, Palladium, or Nickel-Molybdenum) for continuous hydrogenation. This dual-action pathway suppresses coke formation and drives the production of premium, low-sulfur middle distillates, particularly high-quality aviation turbine fuel (jet fuel) and ultra-low-sulfur diesel.
- Aromatics Conversion (Isomerization and Alkylation): In the petrochemical production of polymer precursors, precise shape selectivity is paramount. ZSM-5, with its highly defined medium-pore topology, is heavily utilized for xylene isomerization (maximizing the yield of para-xylene used for polyester manufacturing) and toluene disproportionation. Furthermore, zeolite catalysts dictate the precise alkylation of benzene with ethylene or propylene to yield ethylbenzene and cumene, which are the fundamental building blocks for polystyrene and polycarbonate plastics.
- Light Hydrocarbon Isomerization: To meet stringent environmental regulations requiring the phase-out of toxic gasoline additives (like tetraethyl lead and MTBE), refineries utilize zeolites (such as Mordenite) to isomerize linear light alkanes (like normal pentane and normal hexane) into branched isomers. These branched alkanes possess significantly higher research octane numbers (RON), facilitating the formulation of clean, high-performance gasoline blends.
Coal and Natural Gas Chemistry (C1 Chemistry)
- Methanol-to-Olefins (MTO) / Methanol-to-Propylene (MTP): As the global chemical industry diversifies its feedstock away from crude oil, C1 chemistry has surged. The MTO process utilizes methanol derived from coal gasification or natural gas. This process relies heavily on catalysts with unique cavity architectures, specifically SAPO-34 (a silicoaluminophosphate with CHA topology) or ZSM-5. The large internal chabazite cages of SAPO-34 provide ample space for the "hydrocarbon pool" mechanism to occur, where methanol forms complex polymethylbenzene intermediates. Crucially, the small 8-ring pore openings (approx. 0.38 nm) act as strict molecular turnstiles, allowing only light olefins like ethylene and propylene to escape, while trapping larger aromatics, ensuring an exceptionally high product selectivity for polymer-grade raw materials.
- Methanol-to-Gasoline (MTG) and Methanol-to-Aromatics (MTA): For regions prioritizing energy independence and strategic fuel reserves, zeolites facilitate the direct upgrading of methanol into premium liquid fuels. The MTG process, originally commercialized using ZSM-5, seamlessly converts methanol into a complex mixture of branched alkanes and aromatics perfectly mimicking high-octane gasoline. Similarly, MTA processes tune the acidity of the zeolite to maximize the yield of valuable BTX (Benzene, Toluene, Xylene) chemical feedstocks.
Environmental Control and Emission Treatment
- Diesel Engine Exhaust Denitration (NH3-SCR): Modern commercial diesel vehicles are subjected to severe emission standards (such as Euro VI and EPA 2010). To neutralize toxic nitrogen oxides (NOx), the industry universally employs Selective Catalytic Reduction (SCR). Small-pore zeolites, specifically SSZ-13 or SAPO-34, heavily ion-exchanged with Copper (Cu) or Iron (Fe), serve as the catalyst. Under extreme, rapidly fluctuating exhaust temperatures and high moisture environments, Cu-SSZ-13 effectively catalyzes the reaction between injected ammonia (NH3) and NOx, converting them into completely harmless nitrogen gas (N2) and water vapor. The small pores prevent hydrocarbon poisoning of the active copper sites, ensuring long-term durability.
- Volatile Organic Compounds (VOCs) Abatement: In industrial manufacturing, off-gases often contain dilute but highly hazardous VOCs. Hydrophobic high-silica zeolites function first as intense adsorbent concentrators, capturing VOCs from massive volumes of exhaust air. Once concentrated, dual-function zeolite catalysts facilitate catalytic incineration at significantly lower ignition temperatures compared to thermal oxidizers, converting complex solvents completely into carbon dioxide and water with minimized energy expenditure.
- Greenhouse Gas Conversion: Advanced research and early-stage industrial deployments are leveraging modified zeolites to tackle climate change directly. Zeolites provide the robust frameworks necessary for the catalytic combustion of fugitive methane emissions, as well as the emerging pathways for catalytic hydrogenation of captured carbon dioxide (CO2), transforming a liability into value-added chemicals like methanol or dimethyl ether (DME).
Fine Chemicals and Green Chemistry
- Solid Acid Replacement for Liquid Acids: Historically, fine chemical synthesis relied heavily on massive quantities of highly corrosive, highly toxic liquid acids (such as concentrated sulfuric acid or anhydrous hydrofluoric acid). This generated severe metallurgical corrosion issues and catastrophic volumes of hazardous toxic sludge. Environmentally benign zeolite solid acids have fundamentally transformed these processes. They are now widely deployed in critical esterification, acylation, and acetalization reactions. The solid nature of the catalyst allows for simple mechanical separation via filtration, complete elimination of corrosive hazards, and near-zero hazardous liquid waste discharge.
- Precision Synthesis of Chemical Intermediates: In the high-value sectors of pharmaceuticals, agrochemicals, and synthetic fragrances, molecular purity is paramount. The rigid, shape-selective pores of zeolite catalysts enforce strict steric control over reaction pathways. This capability allows chemists to synthesize highly specific structural isomers or enantiomers required for drug intermediates, completely avoiding the thermodynamic formation of unwanted, closely related byproducts that are notoriously difficult to separate via distillation.
Strategic Selection Criteria for Zeolite Catalysts
Selecting the optimal zeolite catalyst is a rigorous exercise that requires a two-step engineering approach: first, evaluating the foundational physical and chemical boundaries, and second, mapping those capabilities to specific industrial scenarios.
Step 1: The Foundational Screening Funnel
Before addressing specific applications, engineers must filter catalysts through four baseline technical parameters:
- Kinetic Diameter vs. Pore Size: Ensure steric compatibility. The zeolite's pore aperture must be large enough to allow reactants in and products out, but restrictive enough to prevent the formation of bulky, unwanted byproducts.
- Acid Strength and Density: Match the acidity to the reaction's activation energy. Deep cracking requires highly dense, strong Brønsted acid sites, while delicate isomerization demands moderate acidity to prevent over-cracking.
- Operating Environment and Si/Al Ratio: For processes involving high temperatures and steam, specify high-silica zeolites (high Si/Al ratio) to guarantee extreme hydrothermal stability and prevent framework collapse.
- Reactor Type and Mechanical Strength: Specify high-attrition-resistant microspheres for turbulent fluidized-bed reactors, and high-crush-strength extrudates for fixed-bed systems.
Step 2: Scenario-Based Selection Blueprints
Once the foundational boundaries are established, procurement strategies should rely on scenario-based blueprints. Below are the optimal selection frameworks for four demanding industrial environments:
Scenario 1: Heavy Oil Fluid Catalytic Cracking (FCC)
- Core Engineering Challenge: The feedstock consists of massive hydrocarbon molecules. The catalyst operates in a violent, high-velocity fluidized bed and faces extreme high-temperature steam during regeneration.
- Selection Blueprint: Specify a large-pore architecture (e.g., Zeolite Y). It must be deeply dealuminated into an Ultra-Stable Y (USY) structure (high Si/Al ratio) to survive hydrothermal collapse, and formulated into 60–75 micron high-attrition-resistant microspheres.
Scenario 2: Methanol-to-Olefins (MTO) / C1 Chemistry
- Core Engineering Challenge: Demands extreme product selectivity (maximizing ethylene/propylene while blocking aromatics) and faces rapid coking deactivation due to the "hydrocarbon pool" mechanism.
- Selection Blueprint: Avoid large pores. Specify small-pore zeolites with specific cage structures (e.g., SAPO-34) or medium-pore ZSM-5 to act as strict molecular turnstiles. The acid density must be precisely reduced to delay excessive coke polymerization.
Scenario 3: Diesel Exhaust Denitration (NH3-SCR)
- Core Engineering Challenge: The exhaust environment experiences drastic temperature fluctuations, high moisture, and unburned hydrocarbons (HC) that can poison active sites.
- Selection Blueprint: Utilize small-pore zeolites (e.g., SSZ-13 or SAPO-34) to physically block hydrocarbon poisons. The framework must undergo transition metal ion exchange (Copper or Iron) to act as active redox centers for NOx neutralization.
Scenario 4: VOCs Abatement & Catalytic Combustion
- Core Engineering Challenge: Industrial off-gases feature low VOC concentrations mixed with extreme humidity, causing water molecules to competitively occupy traditional adsorbent pores.
- Selection Blueprint: Strong acidity is unnecessary; extreme hydrophobicity is the priority. Specify high-silica or all-silica zeolites. The lack of framework aluminum allows the catalyst to preferentially adsorb organic VOCs even in moisture-laden streams for efficient downstream oxidation.
Technical Bottlenecks and Engineered Solutions in Application
Despite their unparalleled theoretical capabilities, the deployment of zeolite catalysts in harsh industrial realities exposes severe physical and chemical limitations. Acknowledging these bottlenecks and deploying advanced engineering solutions is the hallmark of professional catalyst management.
Physical and Structural Limitations
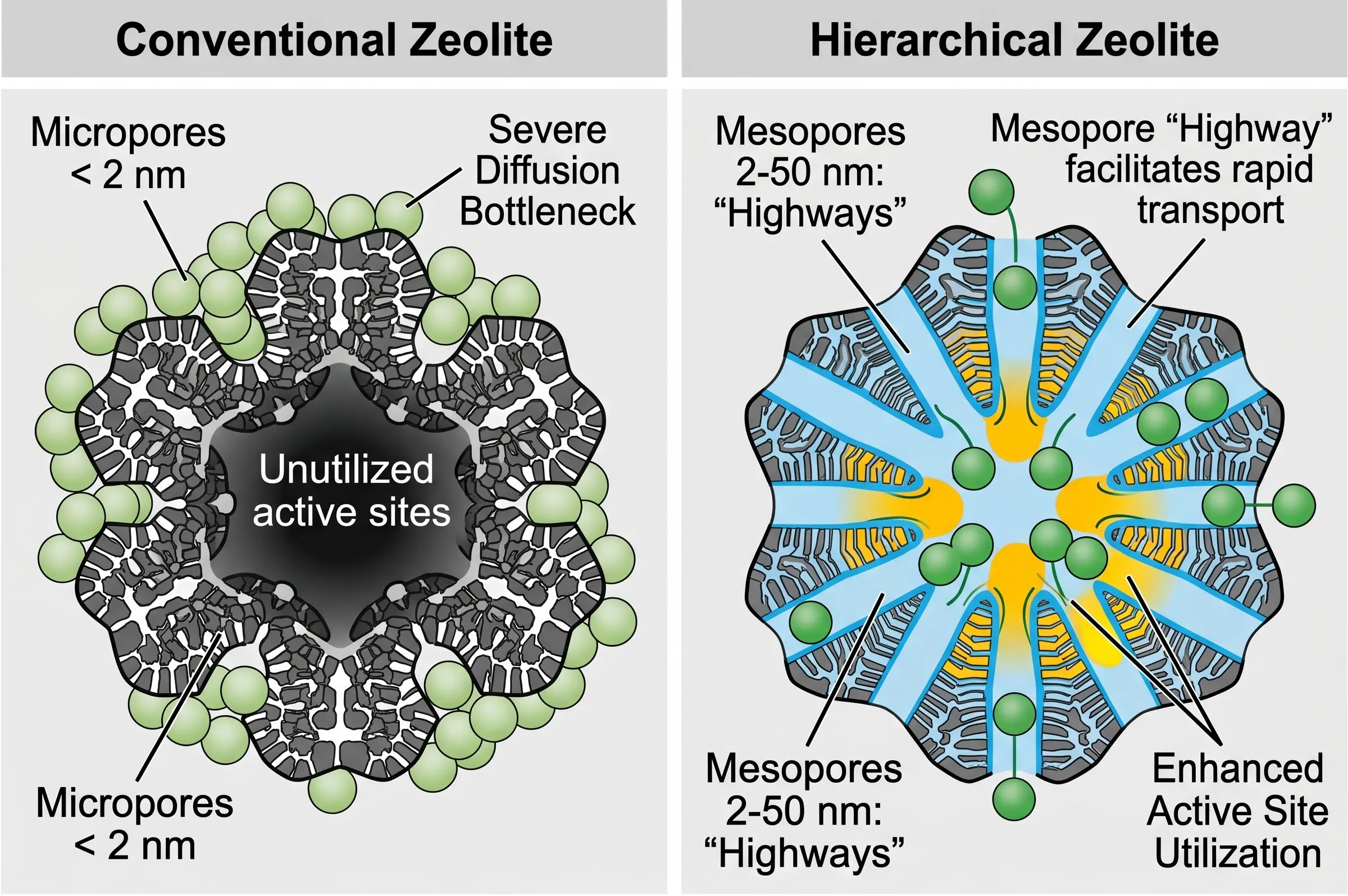
- Mass Transfer Resistance (Internal Diffusion Limits): This is the most profound inherent flaw of purely microporous zeolites. Because the micropore dimensions (e.g., 0.5 nm) are so close to the kinetic diameter of the reactant molecules, diffusion shifts from rapid bulk diffusion to extremely slow Knudsen diffusion, or even single-file diffusion. Molecules enter and exit the pores at an agonizingly slow rate. Consequently, the vast majority of the internal active sites (often over 90%) deep within the crystal remain starved of reactants and are entirely wasted. Furthermore, trapped product molecules undergo secondary reactions, ruining product selectivity.
Engineered Solution: The industry resolves this via the synthesis of hierarchical zeolites (mesoporous zeolites). By chemically etching or utilizing secondary templating agents during synthesis, larger mesopores (2 to 50 nm) are introduced into the crystal. These mesopores act as molecular "highways," allowing bulk reactants to rapidly bypass the bulk crystal and access the microporous "local roads" deeply, slashing diffusion path lengths and unlocking the full catalytic inventory. Alternatively, synthesizing nano-sized zeolite crystals achieves a similar reduction in mass transfer resistance.
-
 Industrial Forming Side-Effects: As established, raw zeolite powder must be bound with inorganic binders (alumina, silica, kaolin) comprising 20% to 50% of the final catalyst weight to achieve physical strength. This binder severely dilutes the concentration of active zeolite. Worse, the binder itself may possess uncontrolled, non-selective acidic sites that drive unwanted side reactions, or the binder material may physically smear over and block the entrances to the zeolite micropores during extrusion, nullifying shape selectivity.
Industrial Forming Side-Effects: As established, raw zeolite powder must be bound with inorganic binders (alumina, silica, kaolin) comprising 20% to 50% of the final catalyst weight to achieve physical strength. This binder severely dilutes the concentration of active zeolite. Worse, the binder itself may possess uncontrolled, non-selective acidic sites that drive unwanted side reactions, or the binder material may physically smear over and block the entrances to the zeolite micropores during extrusion, nullifying shape selectivity.
Engineered Solution: Catalyst manufacturers utilize binder-free forming technologies, where the binder itself is chemically converted into active zeolite in-situ after extrusion. Alternatively, active matrix technologies are employed, where the binder is specifically engineered with large-pore meso-acidity to pre-crack massive molecules before they reach the precise zeolite micropores.
Chemical Deactivation and Extreme Conditions
- Coking Deactivation: This is the inevitable operational nightmare for hydrocarbon processing. Inside the restrictive confines of the micropores, highly reactive olefins and aromatics often become sterically trapped. Under the influence of strong internal Brønsted acids, these trapped molecules undergo rapid, successive polycondensation and cyclization reactions. They fuse into massive, dense carbonaceous polymers—commonly referred to as "coke." This coke acts like molecular concrete, completely sealing the pore channels and suffocating the active sites.
Engineered Solution: Engineers combat coking by prioritizing zeolites with 3D intersecting pore structures (like ZSM-5) that lack spatial "dead ends" where molecules can stagnate. Additionally, precise passivation of external surface acid sites and careful tuning of the internal acid density prevents the excessive sequential reactions that lead to rapid coke formation.
- Hydrothermal Framework Collapse (Dealumination): Industrial reactors are hot, and steam is frequently present (from combustion byproducts, process steam stripping, or oxidative regeneration). Under the aggressive attack of high-temperature steam, the Al-O-Si bonds within the framework undergo hydrolysis. Aluminum atoms are forcibly ejected from the crystalline lattice (dealumination). Once the aluminum is lost, the associated active acid site vanishes permanently. In severe cases, massive dealumination causes the entire crystalline framework to structurally collapse into an amorphous, inactive state. This degradation is highly irreversible.
Engineered Solution: To prevent fatal collapse, manufacturers subject the raw zeolite to severe, controlled hydrothermal treatments and chemical dealumination using chelating agents or steam-calcination (creating USY). This process intentionally removes vulnerable aluminum and allows silicon atoms to migrate and heal the framework defects, creating a highly siliceous, ultra-stable lattice that can endure years of extreme industrial punishment.
- Extreme Sensitivity to Catalytic Poisons: Industrial feedstocks (crude oil, raw coal gas) are laden with contaminants. Because zeolites act as solid acids, any basic compounds in the feed (ammonia, organic amines, or trace sodium ions) will aggressively neutralize the active acid sites, causing immediate catalytic death. More dangerously, heavy metals present in crude oils are fatal. Nickel (Ni) deposits on the zeolite and acts as an aggressive dehydrogenation catalyst, generating massive amounts of unwanted hydrogen gas and accelerating coke formation. Vanadium (V) is catastrophic; at reactor temperatures, it forms highly mobile vanadic acid which physically melts and destroys the zeolite crystal structure.
Engineered Solution: Refineries must deploy rigorous upstream hydrotreating to remove basic nitrogen and metals. Within the catalyst itself, manufacturers incorporate sophisticated passivators. Antimony (Sb) or Bismuth (Bi) compounds are added to aggressively bind with Nickel, preventing its dehydrogenation activity, while rare-earth elements or specialized alkaline-earth traps are incorporated to immobilize Vanadium before it can attack the zeolite framework.
Lifecycle Management and Catalyst Regeneration
A zeolite catalyst is a massive capital expenditure. Maximizing its operational lifespan through rigorous lifecycle management and controlled regeneration is essential for maintaining plant profitability.
- The Regeneration Mechanism: When a zeolite catalyst eventually succumbs to heavy coking, its activity drops below economically viable thresholds. However, unlike irreversible poisoning, coke deactivation can be reversed. The industrial regeneration process involves taking the catalyst offline (or circulating it to a regenerator vessel in fluidized systems) and subjecting it to controlled oxidative combustion—commonly known as "carbon burn-off." By introducing air or an oxygen-nitrogen mixture at elevated temperatures (typically between 500°C and 700°C), the heavy carbonaceous deposits are oxidized and burned away as carbon monoxide and carbon dioxide, successfully unblocking the micropores and restoring access to the acid sites.
- Critical Temperature Control Parameters: The combustion of coke is a violently exothermic reaction. If the oxygen concentration is too high or the gas flow is insufficient to remove the generated heat, the localized temperature within the catalyst bed will skyrocket, resulting in a thermal runaway. If temperatures breach the catalyst's thermal tolerance threshold in the presence of combustion-generated water vapor, catastrophic hydrothermal dealumination and framework collapse will occur instantly. Therefore, regeneration demands extreme precision in controlling oxygen partial pressures and utilizing complex multi-stage heating profiles to ensure the burn-off proceeds smoothly without destroying the lattice structure.
- Total Cost of Ownership (TCO) Evaluation: Procurement based solely on the initial price per kilogram is an engineering fallacy. A comprehensive TCO model must be utilized. Engineers calculate the initial capital cost, the energy expenditure required for multiple high-temperature regeneration cycles, the inevitable percentage of irreversible activity loss following each burn-off (the aging rate), and the ultimate operational lifespan before total catalyst replacement is necessary. A premium, hydrothermally robust zeolite catalyst may carry a higher initial price tag, but by enduring twice as many regeneration cycles without framework collapse, it significantly slashes reactor downtime, reduces fresh makeup rates, and delivers a substantially lower long-term cost to the enterprise.
Partner with JALON for Deep Customization
After strictly defining the required kinetic diameters, acid densities, and hydrothermal resilience parameters for your specific reactor, partnering with a source manufacturer capable of deep, molecular-level customization is paramount. JALON has cultivated 0 years of profound expertise in zeolite engineering, backed by a formidable total annual production capacity of 0 tons for formed molecular sieves and fully automated DCS manufacturing lines. We specialize in supplying high-performance zeolite powders, including the highly specified ZSM-5 topology. To ensure perfect alignment with your reactor physics, JALON supports comprehensive, full-parameter customization encompassing extreme Si/Al ratios (from 2 to ∞), precisely controlled crystal dimensions (D50=0.5–10μm), and diverse specific cation exchanges.









