Catalyst Support: A Guide to Materials and Their Uses
What Is Catalyst Support?
Active chemicals used in industrial applications such as platinum, palladium, and rhodium are very expensive. If these active metals are introduced into a reactor in bulk, they tend to agglomerate into larger catalyst particles. This reduces the reaction rate, resulting in a loss of active substance.
A catalyst support is a solid, highly porous material designed to provide a vast physical area over which these active components (metal nanoparticles, down to a single surface atom) can be dispersed. By anchoring these active components in place, supports create and maintain physical separations of metal particles. The solid support material also determines the shape of the catalyst, such as pellets, extrudates, and spheres, so that the catalyst can be loaded into large-scale industrial reactors without causing large pressure drops in the flow of fluids, maintaining a low pressure drop.
To understand the requirement of a support, it is vital to divide catalysis into two main methods or types: homogeneous and heterogeneous.
Homogeneous Catalysis
The catalyst is in the same phase as the reactants, often functioning as a homogenous solution. The catalyst is more difficult to separate from the reaction mixture, often requiring additional downstream purification steps, which increases process cost.
Heterogeneous Catalysis
The catalyst is in a different phase than the reactants. Typically, the catalyst is a solid, while the reactants are in the gas phase or liquid phase. This state makes it possible to have uninterrupted industrial processes, where the products can exit the reactor while the solid catalyst stays.
The catalyst support is paramount in Heterogeneous catalysis, as it provides the requisite physical state, dimensional stability, and surface geometry to enable sustained, large-scale chemical reactions and various chemical processes.
Key Properties Impacting Catalytic Efficiency
The choice of catalyst support materials is not made randomly. Depending on the industrial processes, engineers must take into account a range of physical and chemical factors in order to ensure that the final catalyst performs to the expectations and maintains the overall activity of the catalyst. Three major factors determine this performance.
Surface Area and Active Site Dispersion
A support’s primary purpose is to maximize the dispersal of the active metal, taking advantage of an increased surface area. Dispersion describes the number of active metal atoms at the surface compared to the total number of metal atoms. Supports with high surface areas (measured as specific surface area in square meters per gram (m²/g) using the BET (Brunauer–Emmett–Teller) method) give greater anchoring opportunities for the active metal. Ultimately, the surface area of a catalyst dictates its potential.
When supports have a surface area of 100 m²/g or even 1000 m²/g, manufacturers can achieve high catalytic activity with smaller loadings of costly noble metals. High dispersion is directly proportional to high turnover frequency (the number of chemical transformations per active site per second). Should process conditions cause surface area to deteriorate, active sites will migrate and merge, resulting in a significant reduction of catalytic activity.
Porosity and Pore Size Distribution
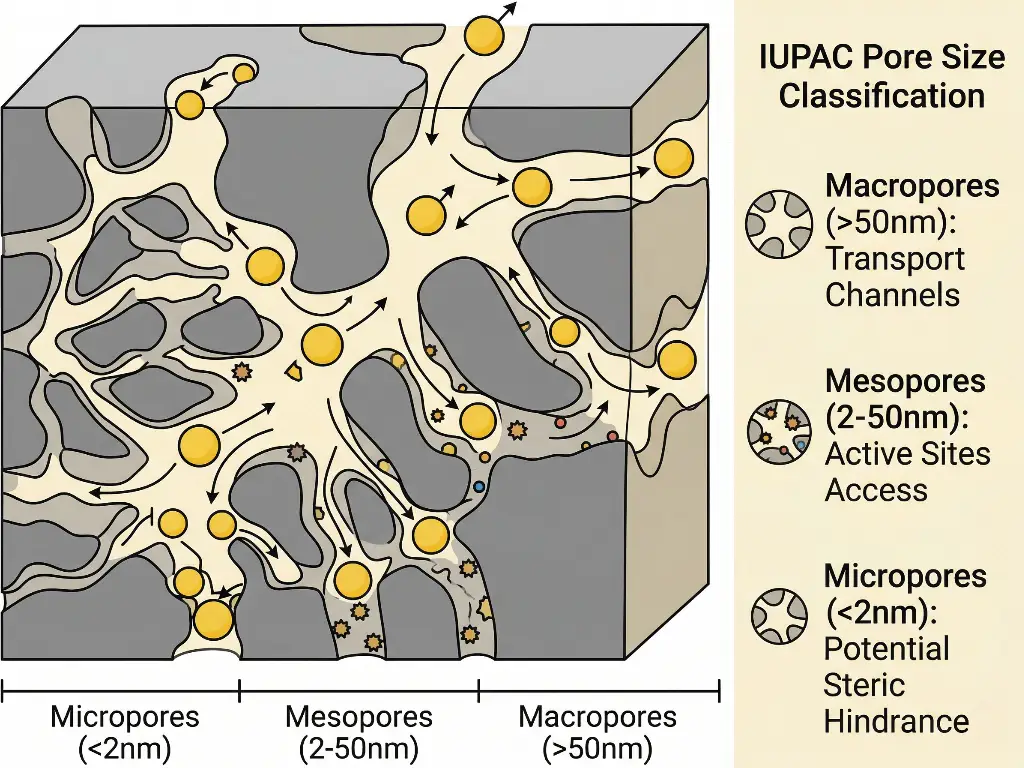
Even with a high surface area, a support structure may be ineffective if its internal active sites cannot be reached by the reactant molecules; the complex pore architecture of a support dictates the mass transfer of a catalyst. Reactants must diffuse through the porous structure, react with the active sites, and the products must diffuse back through the porous structure and exit.
According to IUPAC classification, pore size distribution is divided into three categories:
Micropores
Less than 2 nanometers.
Mesopores
Between 2 and 50 nanometers.
Macropores
Greater than 50 nanometers.
If the pore structure is entirely microporous, and the reactant molecules are large, steric hindrance will make the inner surfaces of the support area inaccessible. On the other hand, if the support is made of only macropores, the diffusion will be fast, but the total surface area and the metal dispersion will be too low to maintain high reaction rates. Therefore, when engineering a catalyst support, the best catalyst supports are those with a combination of pore structures, where macropores are used as transport channels and mesoporous or microporous regions are used to create active sites. This balance guarantees a high pore volume.
Mechanical Strength and Thermal Stability
Chemical processes occur far from the small lab scale beakers with which one might be used to working. Instead, they use large fixed-bed reactors that could be tens of meters tall. A catalyst support has to be able to withstand the hydrostatic pressure of the catalyst bed above it. If the support has inadequate crush strength (lacking sufficient mechanical strength and overall mechanical properties), the bottom-most pellets in the reactor will break apart and turn to dust. This dust accumulates in the empty spaces between the remaining intact pellets, which leads to a large pressure drop in the reactor. Such an event will cause an unplanned and costly shutdown of the entire plant.
Equally important is the thermal stability of the support. Many of the catalytic reactions, including oxidation and hydrocracking, are exothermic. This means that there is a considerable amount of heat generated, and it is important that the support, even in these high temperatures, maintains and preserves its structural identity, exhibiting high thermal stability alongside its pore volume and surface area. If the support material melts, or if it undergoes any phase change or structural collapse due to the elevated temperatures, then that support will also lose catalytic activity.
Industrial Applications of Catalyst Supports
Catalyst supports are built to the specific requirements of different industries. Their specific operating conditions determine which materials are used.
Petrochemical Refining and Processing
Petrochemical refining converts crude oil into fuels and chemical building blocks through processes that operate under extreme temperatures and pressures, often in environments with varying levels of acidity. Notable examples include Fluid Catalytic Cracking (FCC) and catalytic reforming. The supports in these processes must have weak-acidic properties to promote carbon bond cleavage and to initiate isomerization. Because of this, activated alumina and other engineered crystalline molecular sieves are used because they have the necessary acid sites, thermal limits, and mechanical properties to withstand repeated, continuous cycles of carbon regeneration; wherein carbon is burned off at some high temperature.
Environmental Emission Control
Due to the strict regulatory requirements, the control of hazardous emissions, especially volatile organic compounds (VOCs), nitrogen oxides (NOx), and sulfur oxides (SOx), is required. In the process of industrial applications like regenerative catalytic oxidation (RCO) and automotive exhaust, the support facilitates rapid oxidation or reduction reactions. The role of catalyst support here is critical for durability. The treatment of industrial VOCs is made more difficult in high humidity. In these situations, it is often the case that water vapor competes with VOCs for available sites for adsorption along the catalyst. In these cases, hydrophobic (water-repelling) zeolites are used in the treatment of industrial VOCs. These zeolites are uniquely capable of repelling water while selectively adsorbing and concentrating VOCs at active metal sites, ensuring high destruction efficiency in wet exhaust streams.
Fine Chemical Synthesis
Synthesis of pharmaceuticals, agrochemicals, and specialty polymers involves complex, multi-step reactions, such as selective hydrogenations or oxidations. In these processes, high product purity is critical; therefore, side reactions must be strictly avoided, requiring the catalyst support to be chemically inert. If the support has unintended acidic or basic sites, it might catalyze the formation of unwanted byproducts. In fine chemical production, materials with neutral surfaces—such as high-purity silica gel or certain carbon matrices—are commonly employed to achieve absolute selectivity.
Emerging Energy and Electrocatalysis
Electrochemical devices—specifically Proton Exchange Membrane Fuel Cells (PEMFCs) and water electrolyzers for green hydrogen production—are pivotal in the shift toward renewable energy. In these applications, the requirements for catalyst supports differ fundamentally from those in traditional thermocatalysis. Unlike thermocatalytic supports, these materials must be electronically conductive to complete the circuit and engineered to withstand extreme electrochemical potentials in highly acidic or basic liquid phases. Under such harsh conditions, conventional metal oxides tend to dissolve. Consequently, high-surface-area conductive carbons, such as carbon nanotubes, and corrosion-resistant metal oxides (e.g., doped titanium or tantalum oxides) have become the leading choices for electrocatalyst supports.
Common Catalyst Support Materials

For effective engineering specification, it is important to know the intrinsic properties of materials. The following materials exemplify the fundamentals of industrial catalyst support. Each of the materials listed has unique physical and chemical properties which set operational boundaries, largely dictated by their overall chemical stability.
Activated Alumina
γ-Al2O3 (Gamma-alumina) is one of the most widely utilized catalyst supports worldwide. It possesses a unique combination of high surface area (100 to over 300 m²/g), superior mechanical strength, and amphoteric surface characteristics (containing both acidic and basic sites depending on synthesis). Its thermal stability makes it the preferred choice for high-temperature applications, such as petroleum refining, Claus sulfur recovery units, and automotive catalytic converters. Furthermore, its robust physical structure resists high mechanical stress, preventing pressure drops within the reactor. However, at elevated temperatures (typically above 800°C), especially in the presence of steam, gamma-alumina undergoes a phase transition toward α-Al2O3 (alpha-alumina). This structural collapse results in a drastic reduction in surface area and subsequent catalyst deactivation.
Zeolites and Molecular Sieves
Zeolites are crystalline materials made of aluminum and silicon with highly ordered frameworks of internal micropores. Zeolites, unlike amorphous supports, have high uniformity with specific pore sizes of 0.3 to 1.0 nanometers whereas amorphous supports have random, broad distributions of pore sizes. Due to this uniformity and microscale precision, zeolites have a specialized form of catalysis called “shape-selective catalysis”. With these materials, only reactants that are smaller than the pore sizes of the zeolite can access the internal catalytic sites and only specific products can exit and continue to the next stage of the catalysis. In addition to this, zeolites are very desirable because it is possible, by modifying the synthesis of a zeolite and controlling the Si/Al ratio to obtain zeolites with engineered surface acidity from mildly to highly acidic. This characteristic of zeolites makes them extremely valuable in fluid catalytic cracking as well as in complex isomerization.
Silica Gel (SiO2)
Silica gel, an amorphous and highly porous type of silicon dioxide, has an overall neutral surface and yields consistenly over 200 m²/g up to 800 m²/g surface areas. In catalyst manufacturing, the precise control of the internal pore structure throughout the sol-gel synthesis provides the greatest benefit. Furthermore, engineers design silica supports with very specific mesopore and macropore sizes that allow for the passage of large reactant molecules. Due to its entirely inert characteristics, silica is often selected as the support for active metals in cases where acid or base sites would cause undesirable polymerization, cracking, or rearrangements of the reactants. Its greatest drawback is the lack of hydrothermal stability, as the silica framework will degrade when subjected to high temperature and high moisture environments.
Activated Carbon Materials
Different forms of carbon, including activated carbon, carbon black, and engineered carbon nanotubes, have specific surface areas that can exceed 1000 m²/g. The unique structure of carbon enables a number of properties, including intrinsic electrical conductivity, and strong resistance to chemical attacks in highly acidic and highly basic environments. For this reason, carbon serves as the essential substrate for the incorporation of the noble metals (i.e., platinum, palladium, and ruthenium) in liquid-phase hydrogenation, precious metal recovery, and modern electrocatalysis. Nevertheless, carbon supports have a fundamental drawback: a lack of thermal stability in oxidizing environments. When temperatures exceed 400 °C, the carbon support will combust, losing the entire catalyst bed.
Ceramics and Monoliths
When an industrial application requires intense and rapid fluctuations in temperature, gas flow rates, and pressure, the engineering design constraints would render standard particulate supports (pellet and bead supports) ineffective. For these situations, monoliths made of ceramic, especially structures made of cordierite, are used. These monoliths are made of several parallel, straight gas flow channels. Due to cordierite’s low coefficient of thermal expansion, monoliths made of this material are Thermal-shock resistant. Although the bare ceramic monolith has an extremely low surface area (usually < 2 m²/g), a washcoat made of a highly porous material (typically activated alumina) is applied onto the channel walls as a means of increasing the surface area to enhance flow interaction with the washcoat. Subsequently, the active metal is deposited onto this washcoat. This is the typical configuration encountered in automotive catalytic converters and in the emission control systems of large power plants.
Titania and Zirconia
Titanium and Zirconium dioxides (TiO2 and ZrO2) are specialized transition metal oxide supports. Although their baseline surface areas are generally a bit lower than those seen with standard alumina or silica, they are used for a very specific purpose, which is to take advantage of a chemical phenomenon known as Strong Metal-Support Interaction (SMSI). Under certain conditions of a reducing process, some of the surface of these oxides is partially reduced, and the support undergoes a physical migration that partially encapsulates the active metal nanoparticles. This type of physical encapsulation alters dramatically the electronic states and chemisorption characteristics of the active metal, which can result in a more significant enhancement of its catalytic activity and selectivity toward some specific reaction pathways. This is of great significance for industrial photocatalysis, selective CO hydrogenation, and specific syngas conversion.
How to Choose the Right Support
Choosing the correct catalyst support involves matching the exact thermodynamic, chemical stability, and physical conditions of the industrial process to the intrinsic properties of the support material. Using materials “as is” without studying the process conditions invariably leads to catalyst failure. The proper use of catalyst support fundamentally ensures that the catalyst operates efficiently.
The following table presents a summary of the comparative parameters of the main support materials that will be included in the engineering specification process:
| Material Type | Typical Surface Area (m²/g) | Thermal Stability | Surface Acidity/Basicity | Key Industrial Advantage | Primary Limitation |
|---|---|---|---|---|---|
| Activated Alumina | 100 – 300 | Excellent (up to 800°C) | Amphoteric | High crush strength, universal applicability in high-heat processes. | Subject to phase transition (γ to α) at extreme temperatures. |
| Zeolites / Molecular Sieves | 300 – 1000+ | Good to Excellent | Highly Tunable (mostly Acidic) | Precise shape-selectivity, strict pore size control, highly active acid sites. | Susceptible to micropore plugging by heavy hydrocarbons or coking. |
| Silica Gel | 200 – 800 | Moderate | Neutral | Chemically inert, prevents side reactions in fine chemical synthesis. | Lower hydrothermal stability compared to alumina. |
| Activated Carbon | 800 – 1500+ | Poor (in Oxygen) | Neutral / Inert | Excellent chemical resistance to harsh acids/bases, high electrical conductivity. | Combusts in air at elevated temperatures. |
| Ceramic Monoliths | < 2 (requires washcoat) | Exceptional | Neutral | Near-zero thermal expansion, survives massive thermal shocks, low pressure drop. | Requires complex washcoating steps to provide necessary surface area. |
| Titania / Zirconia | 10 – 100 | Good | Mildly Acidic | Exhibits Strong Metal-Support Interaction (SMSI), enhances specific reaction pathways. | Higher material cost, inherently lower baseline surface area. |
To use this data productively, analyze the sector-specific application in a structured manner.
- Determine the Temperature and Atmosphere: For processes above 500°C in an oxygen environment (exhaust gas treatment, for example), carbon components must be ruled out. Activated alumina and cordierite monoliths will be your optimal choices.
- Determine the Need for Selectivity: For reactions requiring separation of molecules differing by a fraction of a nanometer (this includes xylene isomerization and particular gas purifications), zeolite is virtually the standard choice. This is because of the particular crystalline pore sizes.
- Determine the Chemical Environment: For catalysts that work in strongly corrosive liquid acid (as in some electrolyzers), alumina and silica will dissolve. Therefore, the choice is between carbon and very specific metal oxides to ensure a robust structure.
- Determine the Byproduct Risk: For feed materials containing sensitive molecules that tend to polymerize readily at acid sites, avoid using strongly acidic zeolites or amphoteric alumina. Instead, inert silica is recommended to minimize byproduct formation.
Troubleshooting Catalyst Deactivation
Catalysts will is gradually deactivate throughout industrial processes, challenging the integrity of the catalyst support. Even when they initially meet performance specifications, once deactivation occurs, engineers must determine the cause to suggest more adaptable support material to reduce catalyst downtime.
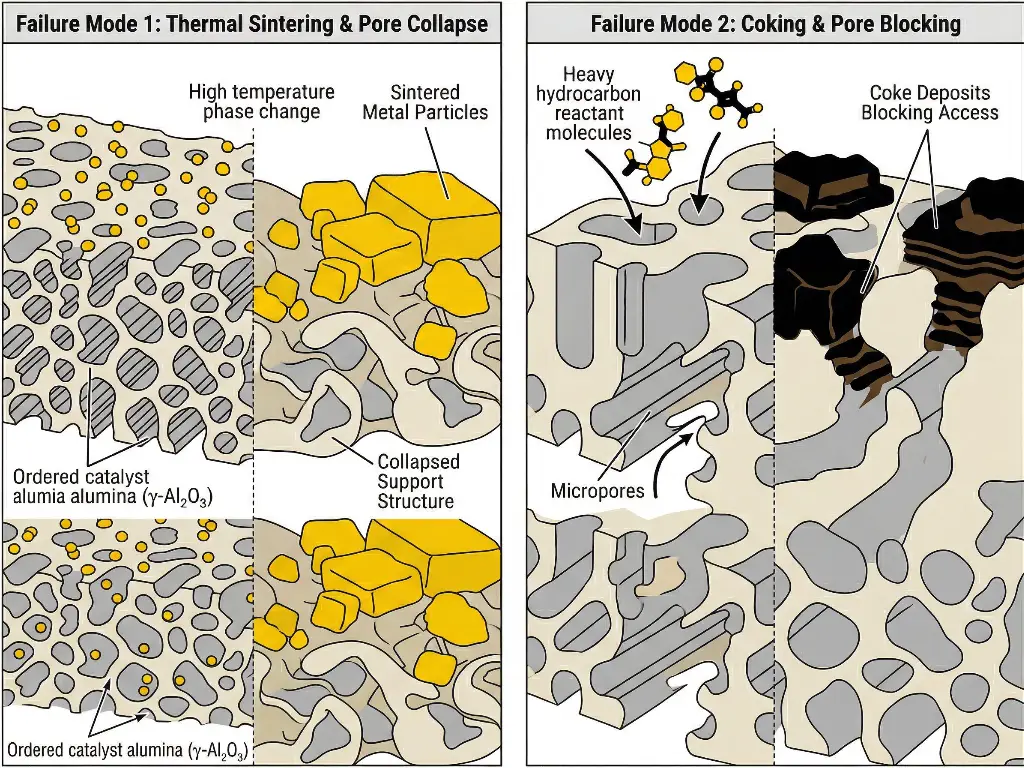
Thermal Sintering and Pore Collapse
Failure occurs when thermal exposures exceed the definite design limits and cause the collapse of the internal pore walls. This results in a drastic microscale collapse of the internal surfaces. This is seen with activated alumina as an irreversible phase change from the high surface area gamma phase to the low surface area alpha phase. As the surface area decreases, the active particles will collapse and agglomerate into large, inactive particles.
Coking and Fouling
During reaction of heavy hydrocarbons, side reactions lead to the formation of coke, which deposits on the internal surfaces of the support. In fully microporous materials, such as classic zeolites, these deposits rapidly obstruct the entrances to the pores, and completely deny the reactants access to the active sites within the crystals.
Active Metal Leaching
The active metal is removed from the support and flows away with the liquid reactant stream. This can happen because the adhesion of the metal to the support is too weak, or the support itself is slowly being dissolved under reaction conditions, carrying the metal along with it.
Conclusion and Next Steps
The specification of a catalyst support is a critical engineering decision that dictates plant efficiency, product purity, and reactor uptime. Whether maximizing the dispersion of platinum, resisting thermal collapse in an oxidation unit, or utilizing the precise shape selectivity of a zeolite framework, the correct physical and chemical properties of the support must be aligned perfectly with the process conditions. Failure to account for factors like pore distribution, crush strength, or surface acidity inevitably leads to rapid coking, sintering, and catastrophic pressure drops.
For operations seeking to optimize their catalytic processes, mitigate deactivation, or develop highly specialized gas separation systems, partnering with an experienced manufacturer is essential. JALON brings 28 years of industry experience, a dedicated team of 91 experts, and a suite of advanced analytical equipment to solve complex adsorption and catalytic challenges. With fully automated production facilities in China and Thailand, JALON provides a secure, globally certified supply chain without Minimum Order Quantities (MOQ).
To evaluate how optimized activated alumina or customized zeolite molecular sieves can improve your process yields, contact our engineering team today for a free project analysis report, custom formulation consultation, and comprehensive sample testing.









